VidyaRA combines “Vidya,” the Sanskrit word for knowledge, with Regulatory Affairs (RA) — reflecting a fundamentals-first, knowledge-led approach to regulatory practice.
VidyaRA is a platform focused on Regulatory Affairs education, technical advisory, and capability building for MedTech professionals around the world.
It was created to address a gap that goes beyond regulations, focussing on how stakeholders understand, apply, and grow with regulatory expectations over time.
Mission
VidyaRA’s mission is to build clarity and confidence in Regulatory Affairs among MedTech professionals worldwide.
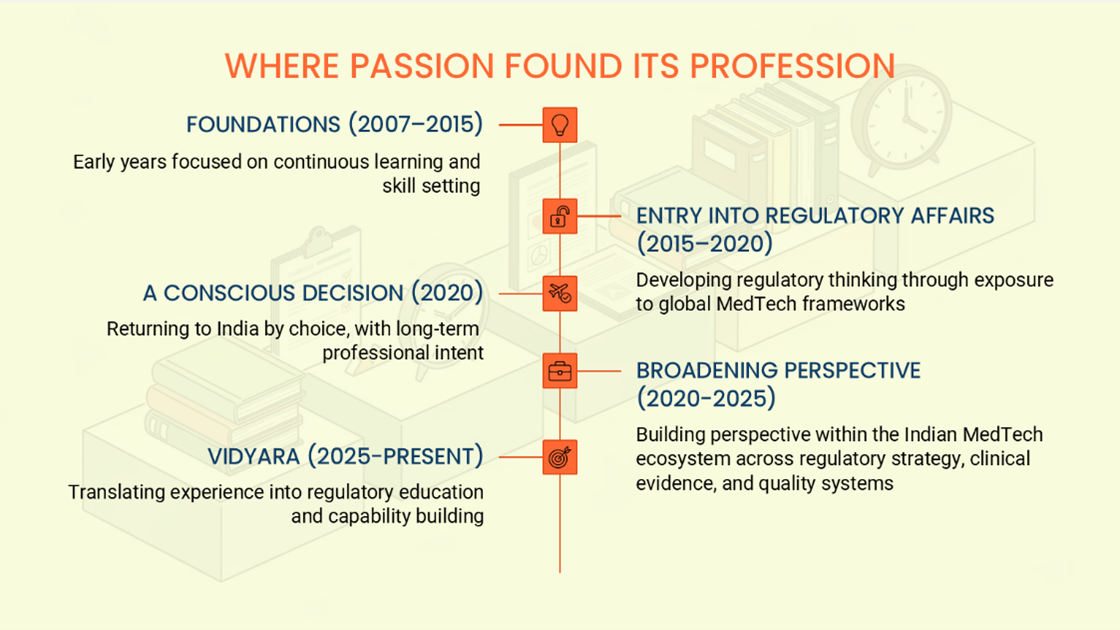
Founder’s Journey
Heena brings over a decade of experience in Regulatory Affairs for medical devices, with exposure across global regulatory frameworks and MedTech operating models. She holds a post-graduate degree in Regulatory Affairs from Northeastern University (Boston, USA).
She began her career with Zimmer Biomet (USA), where she worked extensively on the interpretation and implementation of EU MDR requirements across quality management systems, regulatory, and clinical functions.
Heena made a deliberate decision to return to India, driven by long-term professional intent and interest in the evolving Indian MedTech ecosystem. Since then, her experience has spanned MedTech consulting and manufacturing environments, broadening her perspective across regulatory strategy, clinical evidence, and quality systems.
Her work at VidyaRA is focused on regulatory education and capability building, enabling stronger regulatory thinking for MedTech professionals around the world.
Find her professional credentials below:

RCC-MDR
RAPS, USA

Certified Trainer
ISTD, India

ISO 13485 Lead Auditor
TÜV SÜD, India

ISO 13485 Internal Auditor
DQS, India

ISO 14155:2020
TÜV SÜD, India

Design Controls
Medical Device HQ, Sweden

Usability Engineering
Medical Device HQ, Sweden

Scientific Writing
MeWriT,India


